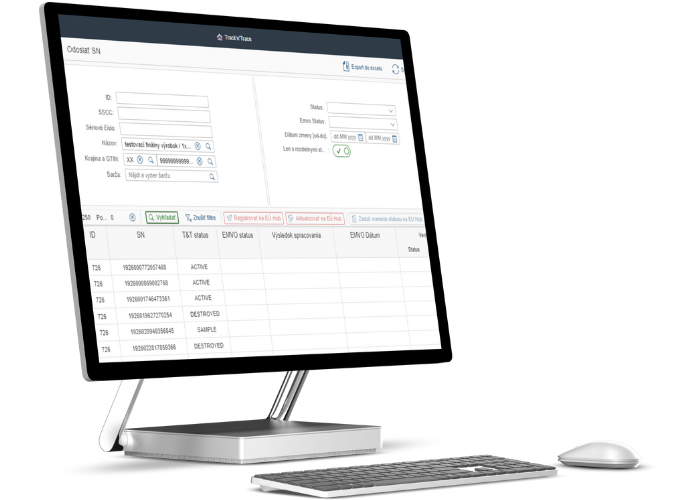
Solution features
- Records of production of medicinal products
- Meets EU regulations
- Security
- Possibility to extend functionality
- Fast implementation
- Integration at EMVO


T&T is a solution that with its functionality, safety and performance will allow to fully label and verify the packaging of medicines on the manufacturer's side, as well as to subsequently control them and unambiguously trace their origin (route in the chain) for the distributor or the end user (patient).
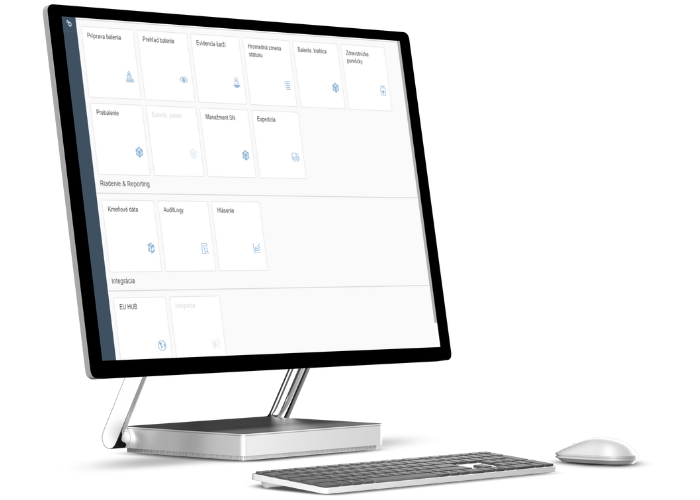
We cover all levels comprehensively, i.e. from the supply and integration of HW and SW into the customer's existing lines, to communication with the MAH (Market Authorisation Holder and EU hub (EMVO).

We continuously monitor current legislative changes and their subsequent incorporation into the products we offer. The new features are also the result of the knowledge and experience of the real-life operation of our product for serialization of medicines, Track&Trace, in practice. Our goal is to continuously improve the efficiency and comfort of our customers' work.